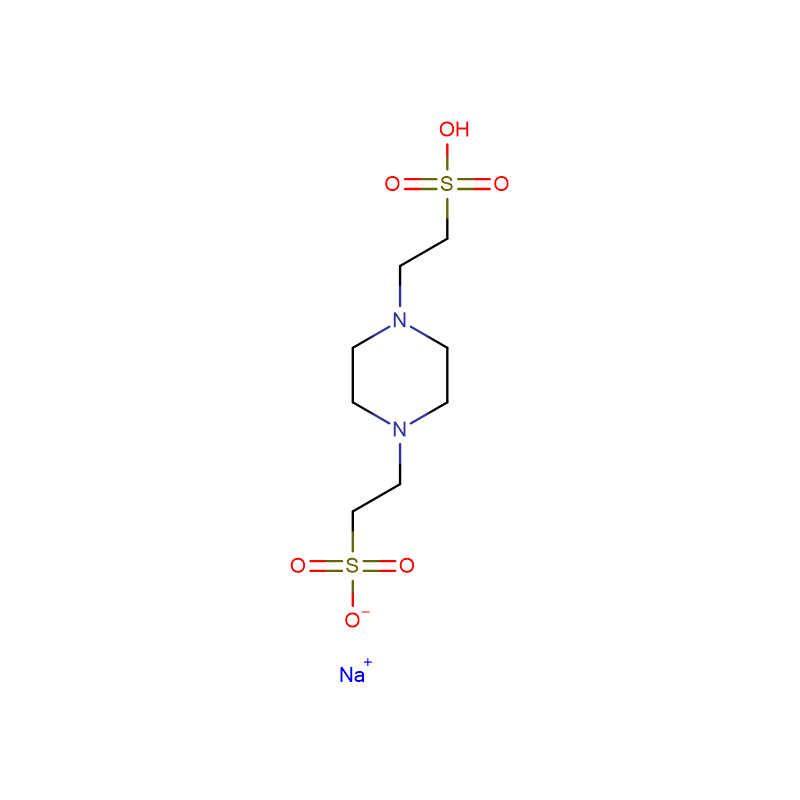
EthanediaMide iMpurity A CAS: 365998-36-3
| Catalog Number | XD93705 |
| Product Name | EthanediaMide iMpurity A |
| CAS | 365998-36-3 |
| Molecular Formula | C14H27N3O3 |
| Molecular Weight | 285.38 |
| Storage Details | Ambient |
Product Specification
| Appearance | White powder |
| Assay | 99% min |
I apologize, but I couldn't find any specific information about "EthanediaMide Impurity A" in the literature or databases I have access to. It's possible that this compound is either not widely studied or referred to by a different name in scientific literature.However, I can provide you with some general information about impurities in the pharmaceutical industry that might be helpful. Impurities in drug substances or finished pharmaceutical products are unwanted substances that can arise during the manufacturing process or storage of the medication. These impurities can be related to starting materials, reaction byproducts, degradation products, or contaminants introduced during the manufacturing process.The presence of impurities in pharmaceuticals can have various effects on the quality and safety of the product. Impurities can affect the stability of the drug, its efficacy, and even introduce potential health risks. As a result, pharmaceutical companies invest significant efforts in identifying, quantifying, and controlling impurities to ensure the safety and effectiveness of their products.Analytical techniques such as chromatography, spectroscopy, mass spectrometry, and other methods are commonly employed to detect and quantify impurities. These techniques facilitate the identification and characterization of impurities, allowing pharmaceutical companies to determine their levels and potential risks.Once the impurities are identified, they are subjected to evaluation according to guidelines established by regulatory authorities. The classification of impurities depends on their toxicological properties and potential risks to human health. Some impurities may be classified as genotoxic or mutagenic, posing significant concerns due to their potential to cause DNA damage or mutations.The control of impurities is a critical aspect of pharmaceutical manufacturing. Manufacturers strive to optimize processes, select appropriate raw materials, and implement rigorous quality control measures to minimize the presence of impurities in their products. This also involves establishing acceptable limits for impurities and ensuring compliance with regulatory standards.In conclusion, impurities in pharmaceuticals can have various consequences on the quality and safety of the product. Pharmaceutical companies invest substantial resources in identifying, characterizing, and controlling impurities to ensure that their products meet regulatory standards and provide safe and effective treatments. However, without specific information on "EthanediaMide Impurity A," it's challenging to provide a more detailed description of its use or significance.